Effective today, several updates have been released in RAMSeS with the key changes highlighted below. For additional information see the full 5.4 release notes.
Enhanced Dashboard Searches
To allow IPF Approvers, PS Project ID Managers, and individuals with Proposal and Award Data Access more robust search capability, additional options have been added to the RAMSeS “Proposals in My Dept” and “Awards in My Dept” dashboard searches.
Export Control IPF screen updates
The current three Export Control IPF screen questions are being replaced by one new required Yes-No question:

If Yes, is answered, a corresponding email will be triggered to the Lead PI (with CC’s to Admin Award/Dept IPF Approvers/PSID Managers):
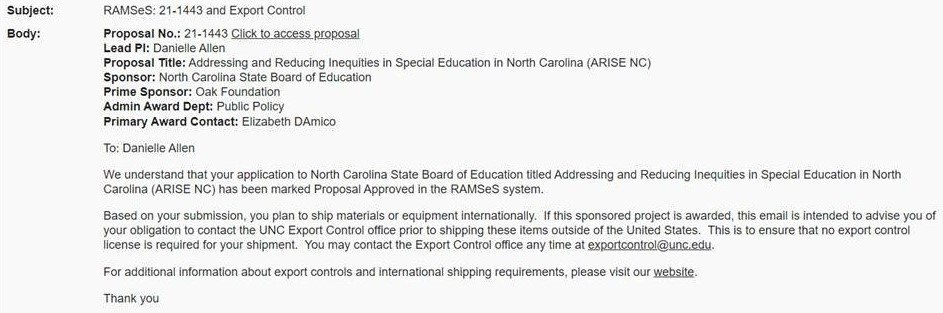
On the Export Control screen for existing proposals in RAMSeS with “No” responses to all of the legacy questions, the new question will be displayed with a “No” response. For existing proposals in RAMSeS with any “Yes” responses, the new question will be displayed with a “Yes” response.
As a reminder, responses to legacy Export Control questions for previously approved proposals may be viewed via the “IPF Approved” snapshot PDF automatically uploaded to the Attachments tab at the time of approval.
Closeout Email Updates
A new clinical trial/ledger 4 paragraph will be included in all 7-, 45-, and 90-day RAMSeS Closeout reminder emails as follows:
“Ledger 4 Clinical Trial Closeouts
If this is a reminder for a ledger 4 clinical trial (4xxxxxx), and the project end date needs to be changed, please complete the Ledger 4 Clinical Trial Date Change Request Form. You will need to select ‘Date Extension’ or ‘Closeout’ as the type of change you are requesting and include documentation that confirms the protocols and study activities are completed.”
*The Corporate and Foundation Relations (CFR) officer (if one has been indicated) will be CC’d on Closeout emails as appropriate.
If you have questions or concerns, please direct them to The Office of Sponsored Research at [email protected] or Sponsored Programs Office at [email protected].